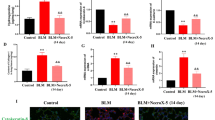
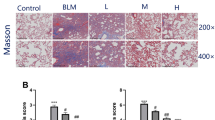
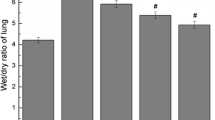
Abstract
Ivermectin is a US Food and Drug Administration (FDA)-approved antiparasitic agent with antiviral and anti-inflammatory properties. Although recent studies reported the possible anti-inflammatory activity of ivermectin in respiratory injuries, its potential therapeutic effect on pulmonary fibrosis (PF) has not been investigated. This study aimed to explore the ability of ivermectin (0.6 mg/kg) to alleviate bleomycin-induced biochemical derangements and histological changes in an experimental PF rat model. This can provide the means to validate the clinical utility of ivermectin as a treatment option for idiopathic PF. The results showed that ivermectin mitigated the bleomycin-evoked pulmonary injury, as manifested by the reduced infiltration of inflammatory cells, as well as decreased the inflammation and fibrosis scores. Intriguingly, ivermectin decreased collagen fiber deposition and suppressed transforming growth factor-β1 (TGF-β1) and fibronectin protein expression, highlighting its anti-fibrotic activity. This study revealed for the first time that ivermectin can suppress the nucleotide-binding oligomerization domain (NOD)-like receptor family pyrin domain-containing protein 3 (NLRP3) inflammasome, as manifested by the reduced gene expression of NLRP3 and the apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), with a subsequent decline in the interleukin-1β (IL-1β) level. In addition, ivermectin inhibited the expression of intracellular nuclear factor-κB (NF-κB) and hypoxia-inducible factor-1α (HIF-1α) proteins along with lowering the oxidative stress and apoptotic markers. Altogether, this study revealed that ivermectin could ameliorate pulmonary inflammation and fibrosis induced by bleomycin. These beneficial effects were mediated, at least partly, via the downregulation of TGF-β1 and fibronectin, as well as the suppression of NLRP3 inflammasome through modulating the expression of HIF-1α and NF-κB.
摘要
伊维菌素是美国食品药品监督管理局(FDA)批准的抗寄生虫剂, 具有抗病毒和抗炎特性. 尽管最近有研究表明伊维菌素在呼吸系统损伤中可能具有抗炎活性, 但其对肺纤维化(PF)的潜在影响尚未得到验证. 本研究旨在探讨伊维菌素(0.6 mg/kg)在PF大鼠模型中减轻博来霉素诱导的生化紊乱和组织学变化的效果. 这可用于验证伊维菌素作为治疗特发性PF在临床上的效用. 在本研究中, 伊维菌素减轻了博来霉素诱发的肺损伤, 表现为炎症细胞浸润减少以及炎症和纤维化评分降低. 值得注意的是, 伊维菌素减少了胶原纤维的沉积并抑制了转化生长因子-β1(TGF-β1)和纤连蛋白的表达, 突显了其抗纤维化活性. 本研究首次揭示伊维菌素可以抑制NOD样受体家族含吡啶结构域蛋白3(NLRP3)炎症小体, 表现为NLRP3和含有半胱天冬酶募集结构域的凋亡相关斑点样蛋白(ASC)基因的表达减少, 以及白细胞介素-1β(IL-1β)水平的下降. 此外, 伊维菌素抑制细胞内核因子-κB(NF-κB)和缺氧诱导因子-1α(HIF-1α)蛋白的表达, 同时降低氧化应激和凋亡标志物的水平. 本研究表明, 伊维菌素可以改善博来霉素引起的肺部炎症和纤维化, 并且部分功能是通过下调TGF-β1和纤连蛋白以及通过调节HIF-1α和NF-κB的表达抑制NLRP3炎性小体来介导的.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ambardar SR, Hightower SL, Huprikar NA, et al., 2021. Post-COVID-19 pulmonary fibrosis: novel sequelae of the current pandemic. J Clin Med, 10(11):2452. https://doi.org/10.3390/jcm10112452
Blobe GC, Schiemann WP, Lodish HF, 2000. Role of transforming growth factor β in human disease. N Engl J Med, 342(18):1350–1358. https://doi.org/10.1056/nejm200005043421807
Buonfrate D, Bisoffi Z, 2021. Standard dose ivermectin for COVID-19. Chest, 159(5):2111–2112. https://doi.org/10.1016/j.chest.2021.03.003
Caly L, Druce JD, Catton MG, et al., 2020. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antiviral Res, 178:104787. https://doi.org/10.1016/j.antiviral.2020.104787
Chen L, Wang T, Wang X, et al., 2009. Blockade of advanced glycation end product formation attenuates bleomycin-induced pulmonary fibrosis in rats. Respir Res, 10:55. https://doi.org/10.1186/1465-9921-10-55
Chen TT, Xiao F, Li N, et al., 2021. Inflammasome as an effective platform for fibrosis therapy. J Inflamm Res, 14:1575–1590. https://doi.org/10.2147/JIR.S304180
Ci XX, Li HY, Yu QL, et al., 2009. Avermectin exerts anti-inflammatory effect by downregulating the nuclear transcription factor kappa-B and mitogen-activated protein kinase activation pathway. Fundam Clin Pharmacol, 23(4): 449–455. https://doi.org/10.1111/j.1472-8206.2009.00684.x
Ding N, Wei B, Fu XH, et al., 2020. Natural products that target the NLRP3 inflammasome to treat fibrosis. Front Pharmacol, 11:591393. https://doi.org/10.3389/fphar.2020.591393
Gad ES, Salama AAA, El-Shafie MF, et al., 2020. The anti-fibrotic and anti-inflammatory potential of bone marrow-derived mesenchymal stem cells and nintedanib in bleomycin-induced lung fibrosis in rats. Inflammation, 43(1):123–134. https://doi.org/10.1007/s10753-019-01101-2
Gazdhar A, Susuri N, Hostettler K, et al., 2013. HGF expressing stem cells in usual interstitial pneumonia originate from the bone marrow and are antifibrotic. PLoS ONE, 8(6): e65453. https://doi.org/10.1371/journal.pone.0065453
Gonçalves K, Vasconcelos A, Barbirato D, et al., 2020. Therapeutic potential of ivermectin for COVID-19. Authorea, preprint. https://doi.org/10.22541/au.159050476.60928563
Han YY, Jiang M, He RL, et al., 2021. Mefunidone ameliorates bleomycin-induced pulmonary fibrosis in mice. Front Pharmacol, 12:713572. https://doi.org/10.3389/fphar.2021.713572
Huang JJ, Xia J, Huang LL, et al., 2019. HIF-1α promotes NLRP3 inflammasome activation in bleomycin-induced acute lung injury. Mol Med Rep, 20(4):3424–3432. https://doi.org/10.3892/mmr.2019.10575
Jiang Q, Geng XK, Warren J, et al., 2020. Hypoxia inducible factor-1α (HIF-1α) mediates NLRP3 inflammasome-dependent-pyroptotic and apoptotic cell death following ischemic stroke. Neuroscience, 448:126–139. https://doi.org/10.1016/j.neuroscience.2020.09.036
Kan XC, Chen YS, Huang BX, et al., 2021. Effect of Palrnatine on lipopolysaccharide-induced acute lung injury by inhibiting activation of the Akt/NF-κB pathway. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 22(11):929–940. https://doi.org/10.1631/jzus.B2000583
Kosyna FK, Nagel M, Kluxen L, et al., 2015. The importin α/β-specific inhibitor ivermectin affects HIF-dependent hypoxia response pathways. Biol Chem, 396(12):1357–1367. https://doi.org/10.1515/hsz-2015-0171
Krolewiecki A, Lifschitz A, Moragas M, et al., 2021. Antiviral effect of high-dose ivermectin in adults with COVID-19: a proof-of-concept randomized trial. eClinicalMedicine, 37:100959. https://doi.org/10.1016/j.eclinm.2021.100959
Latz E, Xiao TS, Stutz A, 2013. Activation and regulation of the inflammasomes. Nat Rev Immunol, 13(6):397–411. https://doi.org/10.1038/nri3452
Lechowicz K, Drożdżal S, Machaj F, et al., 2020. COVID-19: the potential treatment of pulmonary fibrosis associated with SARS-CoV-2 infection. J Clin Med, 9(6):1917. https://doi.org/10.3390/jcm9061917
Lin X, Barravecchia M, Kottmann RM, et al., 2019. Caveolin-1 gene therapy inhibits inflammasome activation to protect from bleomycin-induced pulmonary fibrosis. Sci Rep, 9: 19643. https://doi.org/10.1038/s41598-019-55819-y
Liu Y, Lu FA, Kang LR, et al., 2017. Pirfenidone attenuates bleomycin-induced pulmonary fibrosis in mice by regulating Nrf2/Bach1 equilibrium. BMC Pulm Med, 17:63. https://doi.org/10.1186/s12890-017-0405-7
Mansour SM, Shamma RN, Ahmed KA, et al., 2021. Safety of inhaled ivermectin as a repurposed direct drug for treatment of COVID-19: a preclinical tolerance study. Int Immunopharmacol, 99:108004. https://doi.org/10.1016/j.intimp.2021.108004
Mittal N, Mittal R, 2021. Inhaled route and anti-inflammatory action of ivermectin: do they hold promise in fighting against COVID-19? Med Hypotheses, 146:110364. https://doi.org/10.1016/j.mehy.2020.110364
Raghu G, Brown KK, Collard HR, et al., 2017. Efficacy of simtuzumab versus placebo in patients with idiopathic pulmonary fibrosis: a randomised, double-blind, controlled, phase 2 trial. Lancet Respir Med, 5(1):22–32. https://doi.org/10.1016/S2213-2600(16)30421-0
Rodrigues TS, de Sá KSG, Ishimoto AY, et al., 2021. Inflammasomes are activated in response to SARS-CoV-2 infection and are associated with COVID-19 severity in patients. J Exp Med, 218(3):e20201707. https://doi.org/10.1084/jem.20201707
Ruiz-Riol M, Berdnik D, Llano A, et al., 2017. Identification of interleukin-27 (IL-27)/IL-27 receptor subunit alpha as a critical immune axis for in vivo HIV control. J Virol, 91(16):e00441–17. https://doi.org/10.1128/jvi.00441-17
Sagoo P, Garcia Z, Breart B, et al., 2016. In vivo imaging of inflammasome activation reveals a subcapsular macrophage burst response that mobilizes innate and adaptive immunity. Nat Med, 22(1):64–71. https://doi.org/10.1038/nm.4016
Shin JW, Seol IC, Son CG, 2010. Interpretation of animal dose and human equivalent dose for drug development. J Korean Orient Med, 31(3):1–7.
Sia DK, Mensah KB, Opoku-Agyemang T, et al., 2020. Mechanisms of ivermectin-induced wound healing. BMC Vet Res, 16:397. https://doi.org/10.1186/s12917-020-02612-z
Steinhoff M, Vocanson M, Voegel JJ, et al., 2016. Topical ivermectin 10 mg/g and oral doxycycline 40 mg modified-release: current evidence on the complementary use of anti-inflammatory rosacea treatments. Adv Ther, 33:1481–1501. https://doi.org/10.1007/s12325-016-0380-z
Tian R, Zhu Y, Yao JY, et al., 2017. NLRP3 participates in the regulation of EMT in bleomycin-induced pulmonary fibrosis. Exp Cell Res, 357(2):328–334. https://doi.org/10.1016/j.yexcr.2017.05.028
Vriend J, Reiter RJ, 2016. Melatonin and the von Hippel–Lindau/HIF-1 oxygen sensing mechanism: a review. Biochim Biophys Acta Rev Cancer, 1865(2):176–183. https://doi.org/10.1016/j.bbcan.2016.02.004
Wang JP, Wang H, Fang F, et al., 2021. Danggui Buxue Tang ameliorates bleomycin-induced pulmonary fibrosis by suppressing the TLR4/NLRP3 signaling pathway in rats. Evid Based Complement Alternat Med, 2021:8030143. https://doi.org/10.1155/2021/8030143
Wang ZY, Li XN, Chen H, et al., 2021. Resveratrol alleviates bleomycin-induced pulmonary fibrosis via suppressing HIF-1α and NF-κB expression. Aging, 13(3):4605–4616. https://doi.org/10.18632/aging.202420
Zhang X, Song Y, Ci X, et al., 2008. Ivermectin inhibits LPS-induced production of inflammatory cytokines and improves LPS-induced survival in mice. Inflammation Res, 57(11):524–529. https://doi.org/10.1007/s00011-008-8007-8
Acknowlegment
This work was supported by Open Access funding provided by the Science, Technology & Innovation Funding Authority (STDF) in cooperation with the Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Mai A. ABD-ELMAWLA, Heba R. GHAIAD, and Maha ABDELMONEM: study conception, material preparation, data collection & analysis, and writing; Enas S. GAD and Kawkab A. AHMED: material preparation, data collection & analysis, and writing. All authors wrote the first draft of the manuscript, and they all commented on previous versions of the manuscript. All authors have read and approved the final manuscript, and therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Mai A. ABD-ELMAWLA, Heba R. GHAIAD, Enas S. GAD, Kawkab A. AHMED, and Maha ABDELMONEM declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed. All animals’ procedures were performed in accordance with the Research Ethics Committee of the Faculty of Pharmacy, Cairo University (REC-FOPCU), Egypt (No. BC3203) and with the Helsinki Declaration of 1975, as revised in 2013.
Additional information
Supplementary information
Materials and methods
Supplementary materials and methods
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
About this article
Cite this article
Abd-Elmawla, M.A., Ghaiad, H.R., Gad, E.S. et al. Suppression of NLRP3 inflammasome by ivermectin ameliorates bleomycin-induced pulmonary fibrosis. J. Zhejiang Univ. Sci. B 24, 723–733 (2023). https://doi.org/10.1631/jzus.B2200385
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200385
Key words
- Intra-tracheal instillation
- Immunohistochemistry
- Transforming growth factor-β1 (TGF-β1)
- Nuclear factor-κB (NF-κB)
- Lung fibrosis